|
4/7/2024 0 Comments Whats the density of water
Density differences between water layers determine whether a layer will float or sink in relation to other water layers. To build an understanding of the densities of seawater and fresh water and of how water density differences affect ocean circulation, it is important to understand how temperature and salinity differences form water layers of different densities. The salt in seawater comes from eroding land, volcanic emissions, reactions at the seafloor, and atmospheric deposition. Pure water has its highest density of 1000 kg/m3 at a temperature of 3.98oC (39.2oF). Seawater has unique properties: it is saline, its freezing point is slightly lower than fresh water, its density is slightly higher, its electrical conductivity is much higher, and it is slightly basic. Learn how to calculate the density of water using a formula, a table, and a standard value of 1 gram per milliliter or 1 gram per cubic centimeter. Ocean Literacy Fundamental Concept: Most of Earth’s water (97 percent) is in the ocean. The density of water is the weight of the water per its unit volume, which depends on the temperature of the water. The shape of the ocean basins and adjacent landmasses influence the path of circulation. At 8 degrees Celsius, it has a density of 999. It means water is less denser (the same mass occupies more volume) at zero degrees Celsius than water at 4 degrees Celsius. Ocean Literacy Fundamental Concept: Throughout the ocean there is one interconnected circulation system powered by wind, tides, the force of the earth’s rotation (Coriolis effect), the sun, and water density differences. At 0 degrees Celsius density of water is 999.8 kg/cubic meter Being in liquid form water has a density of 998.8 kg per cubic meter at 0 degrees Celsius. What is the density of water in g/cm3 Students’ answers will vary, but their values should mostly be around 1 g/cm3. At room temperature, around 2025 C, the density is about 0.99 g/cm3. Water is most dense at 4 C and at that temperature has a density of 1 g/cm3. Principle 1: The earth has one big ocean with many features density of water changes with temperature. The relative floating and sinking of water masses affects vertical ocean circulation. For example, fresh river water floats on top of salty ocean water (Fig. The relative density of each water mass determines whether it will float or sink in relation to another water mass.  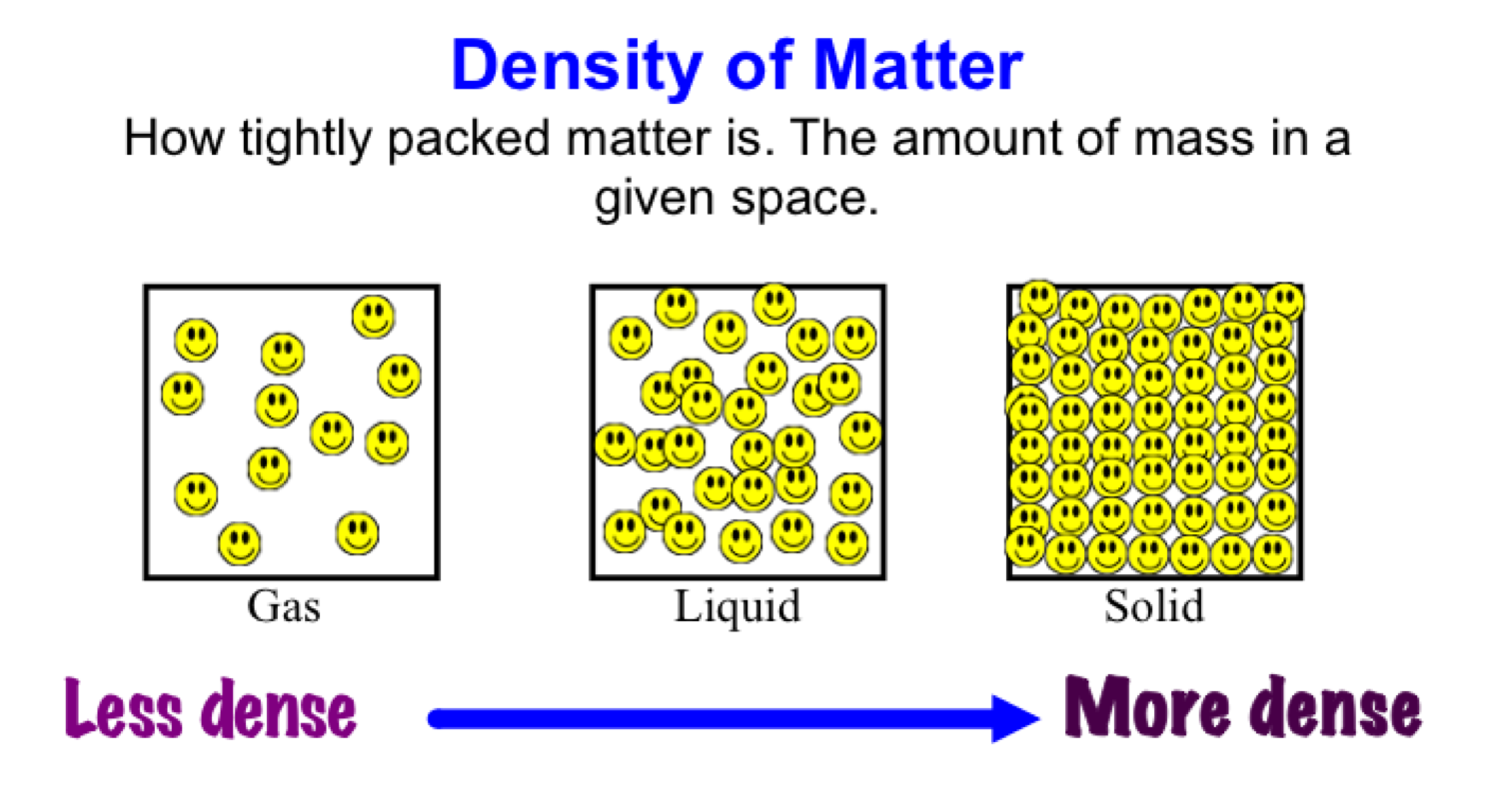
The density of water increases with decreasing temperature, reaching a maximum at 4.0 ° C, 4.0 ° C, and then decreases as the temperature falls below 4.0 ° C 4.0 ° C. Table 14.2 shows the density of water in various phases and temperature. If water masses do not have the same densities, they will form layers of water. The density of solids and liquids normally increase with decreasing temperature. Even within one geographical area of the ocean, there are water masses with different properties. This is why seawater at Antarctica is very salty.Water in the ocean is not uniform in its composition the properties of ocean water vary in different parts of the ocean. The salt that is rejected forms brine beneath the ice and becomes more and more salty until it becomes so dense that it sinks, displaces less dense seawater that moves to the surface. As ice forms in the sea, the salt cannot form part of the ice crystal so the ice is almost pure water.The salinity of seawater is about 3.5% and it freezes at about -1.9☌.The addition of salt to water makes a solution that is denser than fresh water – it freezes at a lower temperature.The hydrogen of the water molecule is attracted to chlorine ions and the oxygen to the sodium ions. This behavior of the density of water explains why ice forms at the top of a body of water. This allows the sodium and chlorine ions to be pulled apart by the water molecules. What is the density of water in grams per millimetre as it is put down in chemistry Using a bathroom scale, a tub, a spoge and a measuring cup explain how you. The density of water increases with decreasing temperature, reaching a maximum at 4.0 ☌, and then decreases as the temperature falls below 4.0 ☌. Salt dissolves in water because the attraction between the water molecules and the sodium ions or chlorine ions is stronger than the attraction between the sodium ions and chloride ions in the lattice.For every sodium ion you will find one chlorine ion (1:1 ratio). Salt is made up of many sodium and chlorine ions stacked together in a lattice.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
